Biomedical Flows Simulation & Multiscale Modeling Lab (BioSiMM Lab)
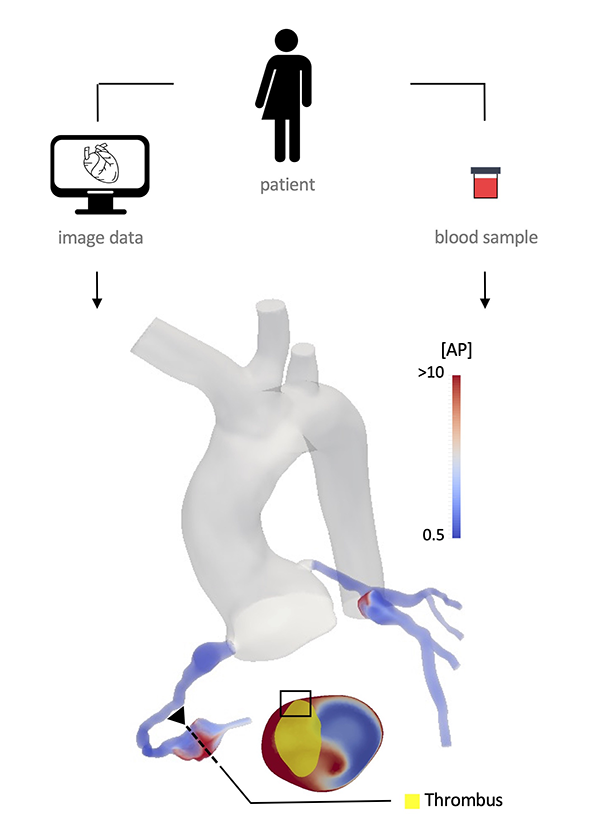
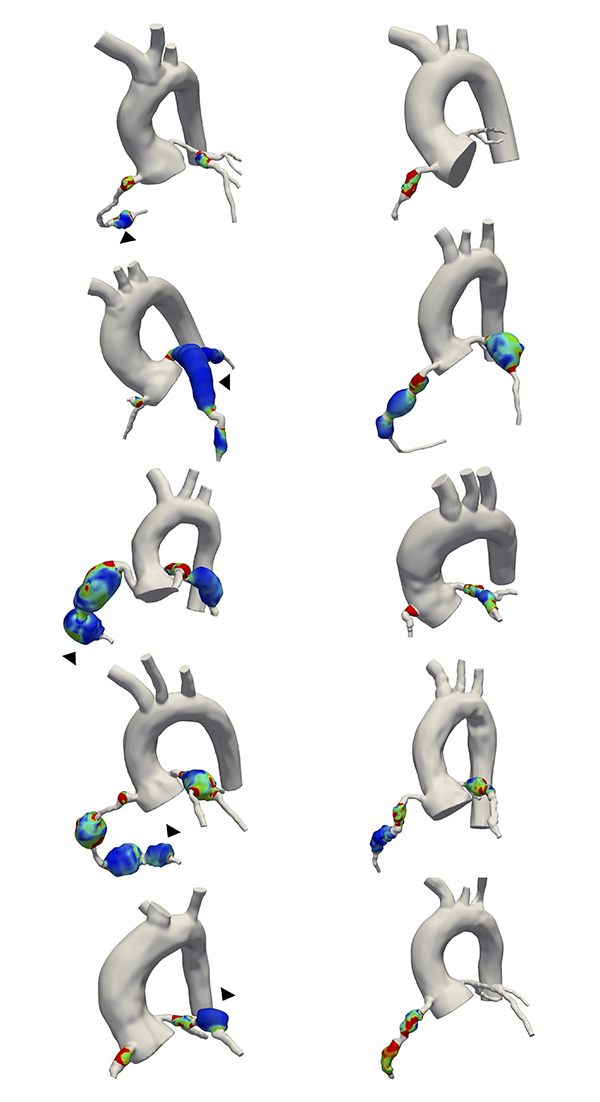
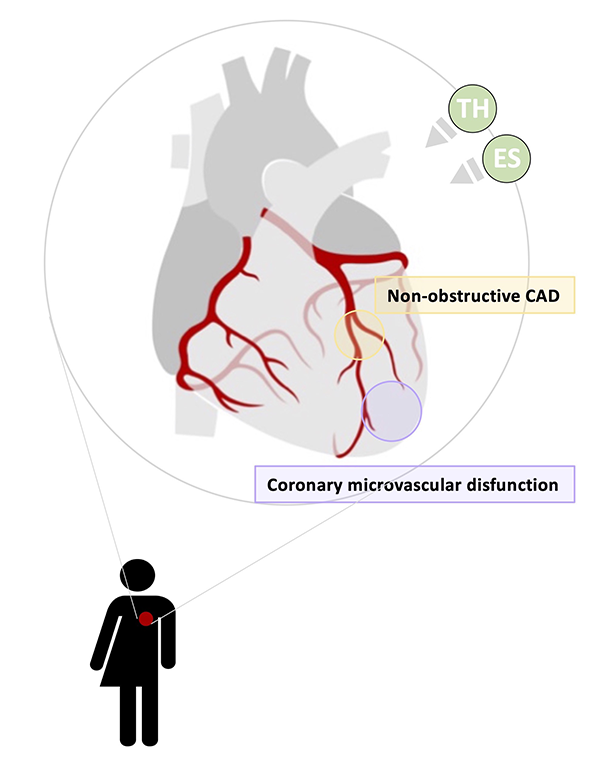
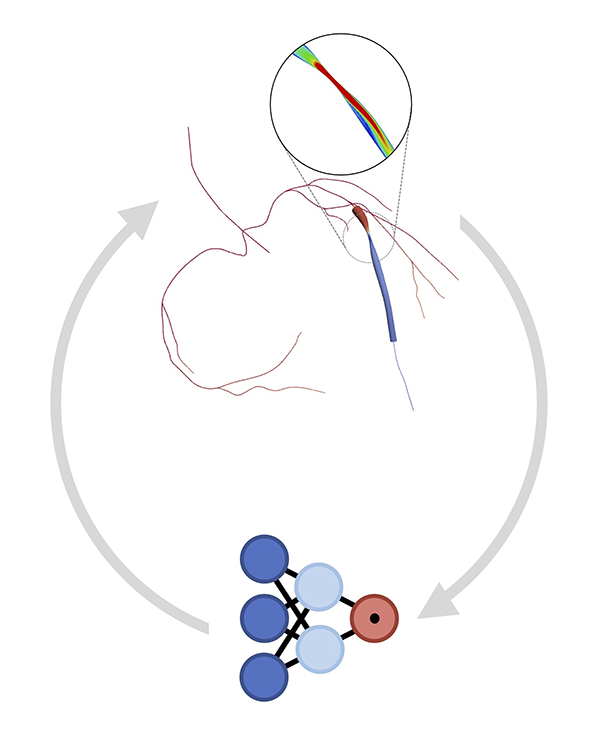
Computational methods are emerging as relevant tools for diagnosing, treating, and quantifying disease progression. However, the translation of these computational methods to the clinic is not yet a reality. The BioSiMM Lab focuses on patient-specific multi-scale modeling and computational fluid dynamics. Our goal is to leverage computational simulations to provide improved patient-specific risk-stratification metrics and enable the design of personalized therapies at a reasonable computational cost. The group's research areas include thrombosis, vascular and microvascular blood flow modeling, transport and drug delivery, and medical device development, emphasizing investigating sex differences in cardiovascular disease.